Community Oncology Practices: Market Trends and Manufacturer Competitive Assessment
Highlights of the report:
Download a PDF of these Highlights
In response to declining reimbursement and other economic challenges, community oncology practices (COPs) are entering 2026 focused on protecting financial viability while selectively expanding key capabilities. HIRC's report, Community Oncology Practices: Market Trends and Manufacturer Competitive Assessment, reviews the market environment and strategic priorities for COPs, and provides a competitive assessment of oncology manufacturers in COP engagement. The report addresses the following questions:
- What are COPs' top market concerns and strategic priorities in 2026?
- What is the status of COP activity related to oncology pharmacy and dispensing services (e.g., alternate site infusion, specialty pharmacy)?
- What is the status of oncology preferred drug lists, clinical pathways, and other utilization management tactics?
- What is the status of oncology biosimilar adoption among COPs?
- Which firms are most often nominated as COPs' Partner of Choice? Which firms are nominated as providing the best oncology-related support offerings?
- How do pharmaceutical firms benchmark in account engagement and quality of oncology account managers, medical/clinical science liaisons, and field-based reimbursement managers?
Key Finding: COP respondents identify optimization of drug purchasing, GPO contracting & inventory management as their top strategic imperative in 2026, followed by optimization of PA, denial management & revenue recovery processes.
Top Market Trends for Community Oncology Practices in 2026. The top disruptive market dynamic/trend for community oncology practice senior leadership in 2026 is Declining Reimbursement & Margin on Pharmaceuticals, followed by Reduction in Medicare Reimbursement, Clinical Workforce Shortages & Recruitment, Impacts of the IRA, and Expiration of Enhanced ACA Subsidies.

The full report provides the complete listing of 20+ trends identified by oncology practices, as well as their top strategic priorities for the next 12-18 months.
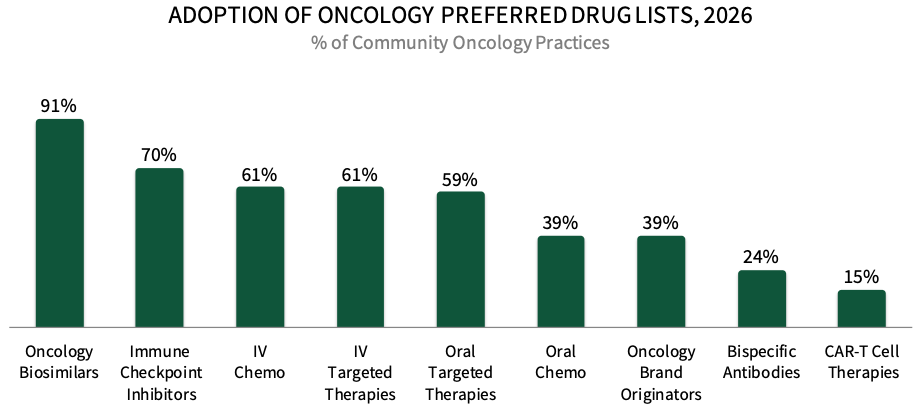
COPs Most Frequently Designate Preferred Products in Categories with Oncology Biosimilars. Over 90% of COP decision-makers in HIRC's sample report having preferred oncology biosimilar products in place in 2026, followed by 70% with preferred immune checkpoint inhibitor medications, 61% with preferred IV conventional chemotherapy products, and 61% with preferred IV targeted therapies. The full report examines biosimilars in detail, including which are preferred, how COPs promote biosimilar use, and which manufacturers offer the best biosimilar support services.
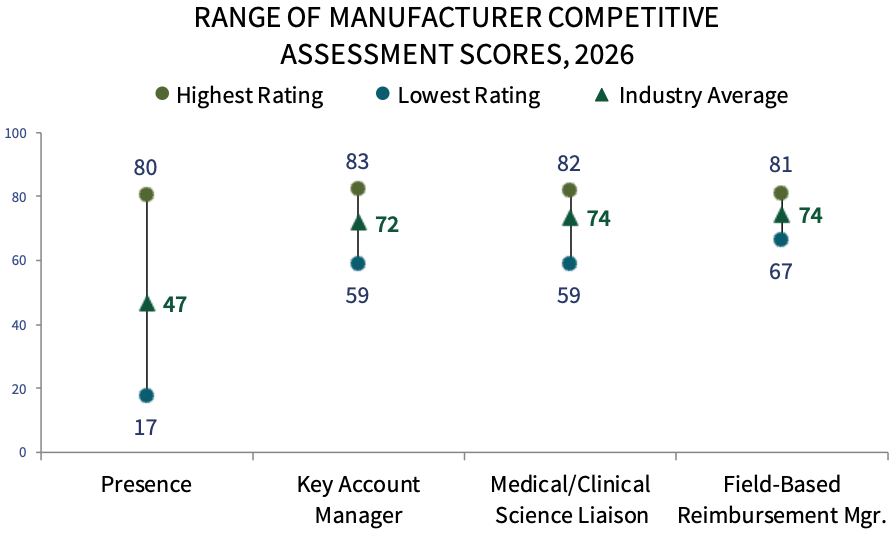
Pharmaceutical Manufacturer Competitive Assessment. Community oncology practice respondents were asked to evaluate three types of customer-facing personnel as noted below. Genentech, Pfizer Oncology, Merck Oncology, Bristol Myers Squibb, and Amgen rate highest in manufacturer field personnel engagement in 2026. The complete report provides COP executives' ratings of 30+ firms active in oncology, as well as ratings of manufacturers' oncology-related support offerings and nominations for overall Partner of Choice.
Research Methodology and Report Availability. In January HIRC surveyed 46 executives from community oncology practices. Online surveys and follow-up telephone interviews were used to gather information. The full report, Community Oncology Practices: Market Trends and Manufacturer Competitive Assessment, is available now to HIRC’s Managed Oncology subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >

