Medicare Advantage Plans: Oncology Medication Management, Contracting, and Manufacturer Engagement
Highlights of the report:
Download a PDF of these Highlights
The evolving policy landscape, driven by the Inflation Reduction Act and emerging Maximum Fair Price regulations, is transforming how Medicare Advantage plans approach contracting and management of oncology medications. HIRC's report, Medicare Advantage Plans: Oncology Medication Management, Contracting, and Manufacturer Engagement, examines management approaches, the contracting environment, and best-in-class manufacturer support in oncology. The report addresses the following questions:
- How are MA plans responding to the Inflation Reduction Act's Part D benefit design changes in the context of oncology drug management? What impact could negotiated Maximum Fair Prices (MFPs) have on the oncology medication contracting landscape?
- What is the status of MA plans' utilization management tactics across oncology medication types?
- What is the status of oncology preferred drug lists (PDL) and clinical pathway adoption across nineteen cancer types?
- How are plans managing oncology medication distribution (e.g., preferred specialty pharmacy, white bagging, site of care management)?
- Which manufacturers stand out as best in Medicare Advantage plan engagement to support their oncology portfolios?
Key Finding: To better manage oncology costs amid Inflation Reduction Act changes, plans report tightening prior authorization rigor and evaluating step therapy where possible.
Contracting Environment for Oral Targeted Cancer Therapies. When asked about the contracting environment for oral targeted therapies, Medicare respondents indicate that combination contracts involving flat access rebates and price protection are most common.
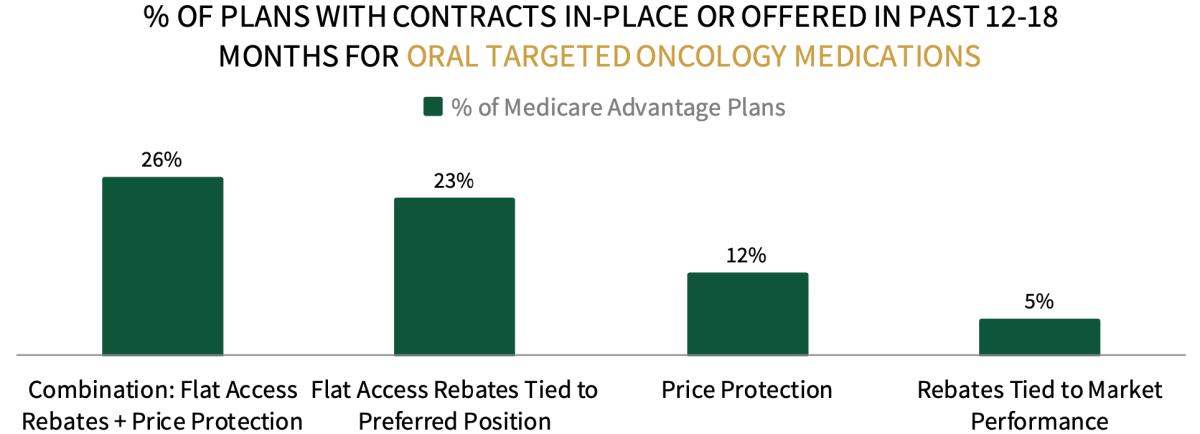
The full report examines contracting approaches and most common discount amounts offered across oncology biosimilars and brand originators, oral and IV targeted therapies, CAR-T cell therapies, immune checkpoint inhibitors, and bispecific antibodies.
Medicare Advantage Plans are Actively Preferring Oncology Medications in 2025. About 44% of respondents in HIRC's sample report that their plan has preferred oral targeted therapies for their Medicare Advantage population in 2025, followed by 35% with preferred IV targeted therapies, 33% with preferred immune checkpoint inhibitors, and 14% with preferred CAR-T cell therapies and bispecific antibodies.
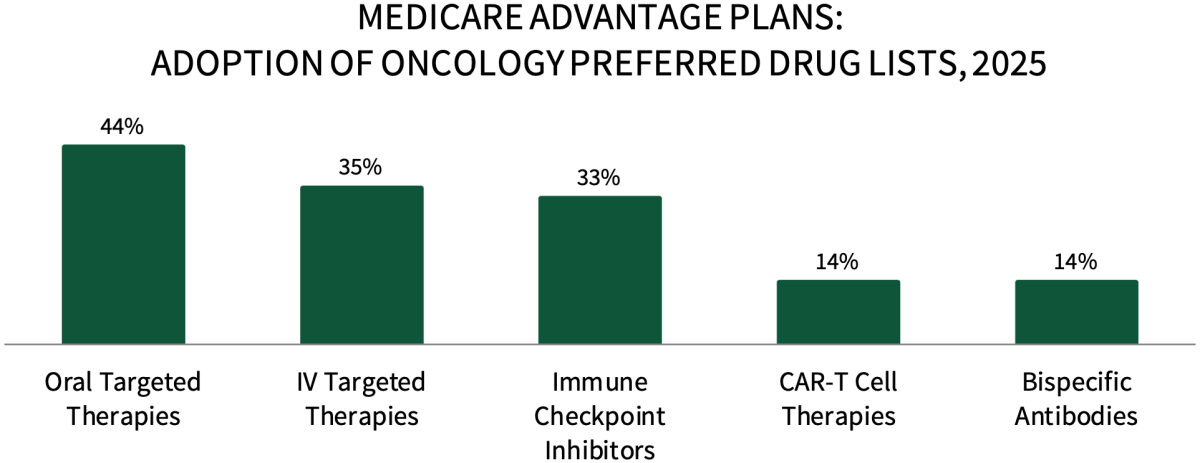
The full report examines the status of preferred products across 19 cancer types, as well as the uptake of oncology biosimilars, and utilization management tactics used to promote preferred products.
Pfizer Oncology Leads as Most Supportive Oncology Manufacturer for Medicare Advantage Plans. Medicare Advantage plan decision-makers were asked to nominate a manufacturer in three key areas: 1) Partner of Choice in Oncology, 2) Best-in-Class Programs/Resources, and 3) Most Willing to Contract for medications covered under Medicare Part D and Part B. Pfizer Oncology and Amgen are consistently among the leaders nominated across best-in-class categories.
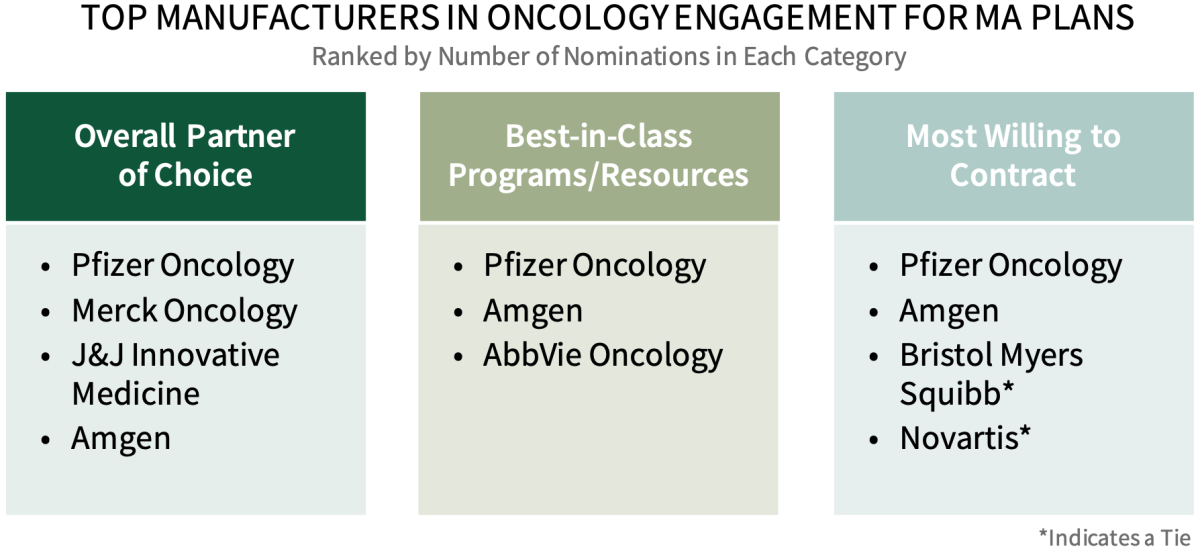
The full report provides additional insights into leading manufacturers' support as well as the factors driving best-in-class nominations.
Research Methodology and Report Availability. In August/September HIRC surveyed 43 pharmacy and medical directors from national, regional, and BCBS plans representing 23 million MA lives. Online surveys and follow-up telephone interviews were used to gather information. The complete report, Medicare Advantage Plans: Oncology Medication Management, Contracting, and Manufacturer Engagement, is available now to HIRC’s Managed Oncology subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >

