Commercial Health Plans: Oncology Medication Management and Market Access
Highlights of the report:
Download a PDF of these Highlights
Commercial MCOs are ramping up bold, proactive strategies to control oncology drug costs and utilization, driven by the surge of groundbreaking therapies entering the market. HIRC's report, Commercial Health Plans: Oncology Medication Management and Market Access, reviews plans' approaches to oncology management and examines the resulting market access landscape. The report addresses the following questions:
- What key oncology-related market trends are commercial health plans currently monitoring?
- What is the status of payers' utilization management tactics across oncology medication types (e.g., targeted therapies, checkpoint inhibitors, biosimilars, CAR-T)?
- What is the status of oncology preferred drug lists (PDL), exclusion lists, and clinical pathway adoption across cancer types?
- How are plans managing oncology medication distribution (e.g., preferred specialty pharmacy, white bagging, site of care management)?
Key Finding: In 2025, health plans are relying most on tighter prior authroization criteria, site of care management for infusions, and promoting the use of biosimilars and generics to control the cost and utilization of oncology medications.
Increased Availability and Uptake of Oncology Biosimilar Medications Among Top Market Trends for Health Plan Leadership. Commercial health plan decision-makers were asked to identify the top oncology-related market trends impacting their business. The top trend identified is the increased availability and uptake of oncology biosimilar medications, followed by increasing cost of medications, and adoption of cell and gene therapies. A greater percentage of plans mentioned the utilization of oncology biosimilars & generics in 2025, compared to 2024.
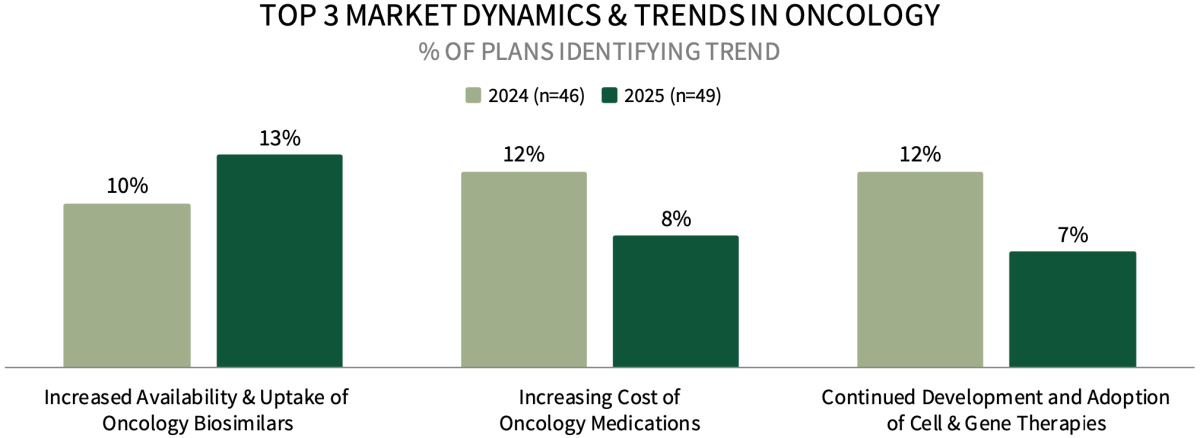
The complete report examines the full listing of 23 market trends identified by health plan leadership.
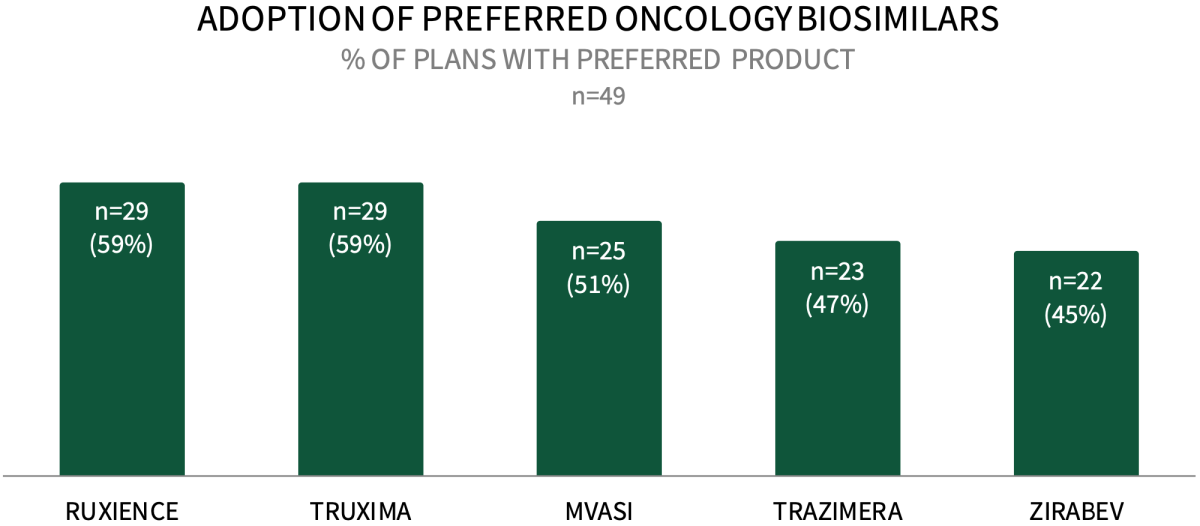
Commercial Payers are Actively Preferring Oncology Biosimilars in 2025. RUXIENCE and TRUXIMA are the most commonly preferred oncology biosimilars among commercial health plans, with roughly 59% of respondents indicating their plan lists these medications as preferred. They are followed by MVASI (51%), TRAZIMERA (47%), and ZIRABEV (45%). The full report examines the status of uptake across 15 oncology biosimilars, and the management tactics used to promote their utilization.
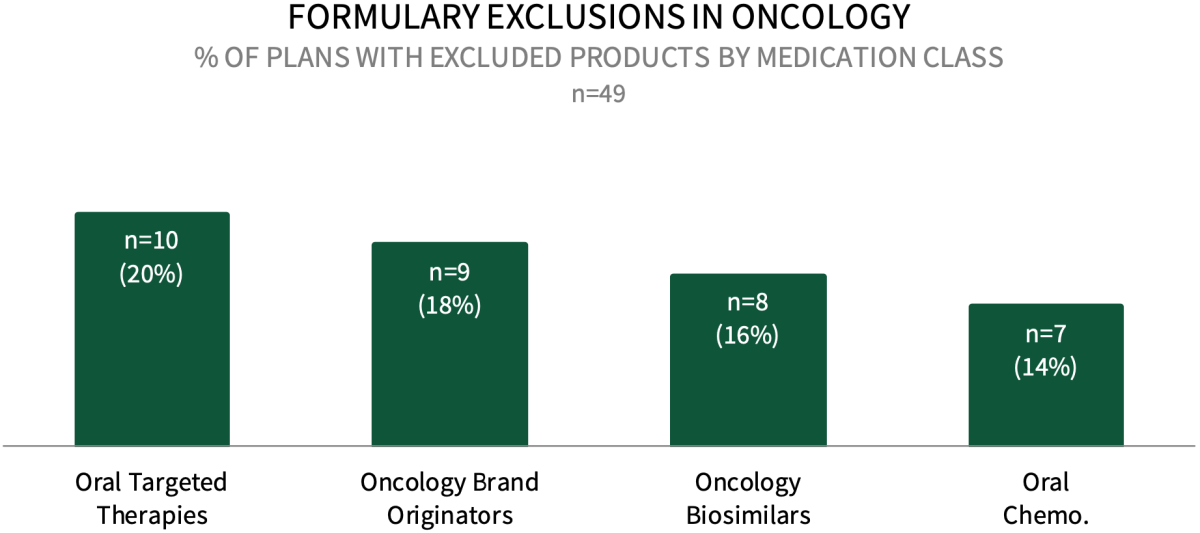
Formulary Exclusions in Oncology. Commercial plans' use of formulary exclusions in oncology remains relatively low. About 20% of commercial health plans say their plan currently exclude some oral targeted cancer therapies from formulary, while another 18% report exclusions for oncology brand originators, 16% for oncology biosimilars, and 14% for oral conventional chemotherapy medications. The full report examines formulary exclusions across nine oncology treatment/therapeutic categories.
Research Methodology and Report Availability. In June 2025, HIRC surveyed 49 pharmacy and medical directors from national, regional, and BCBS plans representing 116 million commercial lives. Online surveys and follow-up telephone interviews were used to gather information. The complete report, Commercial Health Plans: Oncology Medication Management and Market Access, is available now to HIRC’s Managed Oncology subscribers at www.hirc.com.
Download a PDF of these Highlights
Download Full Report (Subscribers only) >

